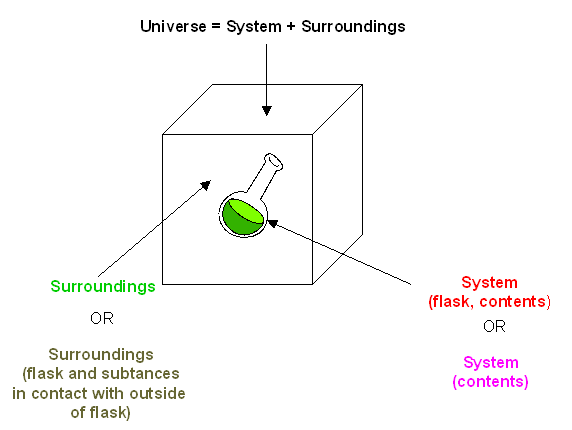
Chemical energy is the energy that is stored in the bonds of all molecules. Whenever we create or break a molecular or ionic bond there is an absorption or release of energy. Outside of the laboratory, we can use this absorption or release of energy to create items for our day to day use. One simple example of this usage is the first aid hot and cold packs. Inside the packs are chemicals contained in separate sections. When the pack is "squashed" and shaken the sections are ruptured and the chemicals mix. If the reaction between the chemicals is exothermic, the liquid in the pack becomes warm. If the reaction is endothermic, the liquid is cooled or in some cases frozen.

Another very common example of how the real world uses the energy of chemical reactions is in the process of photosynthesis.
Photosynthesis is an example of an endothermic chemical reaction. In this process, plants use the energy from the sun to convert carbon dioxide and water into glucose and oxygen. This reaction requires 15MJ of energy (sunlight) for every kilogram of glucose that is produced: