

Experiment 15 Chromatography of Amino Acids |
Introduction/Background
Amino acids are the building blocks of proteins. The amino acid sequence determines the structure and function of the protein. Amino acids are also precursors for other biologically important molecules such as some hormones and neurotransmitters. There are 20 biologically important amino acids. They have at least one amino group (-NH2) and one carboxyl group (-COOH). The general structure for an amino acid is:

The side chain(s) ("R" in the general structure) for each amino acid are different. The liver removes excess amino acids in our bodies. Amino acids are found in all foods except oil. Essential amino acids are those that must come from diet. Our bodies are not able to produce these, and they include isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine. The rest can be made by us, and are non-essential. See the structure of amino acids.
Key Concepts
 alanine structure |
When an amino acid is dissolved in water, it exists in solution as the dipolar ion, or zwitterion. A zwitterion can act as either an acid or a base and has both a positive and negative charge. Substances having this dual nature are amphoteric and are often called ampholytes. An example of the zwitterionic form of alanine is given. |
| Paper chromatography is a method for analyzing mixtures by separating the components in the mixture by their size and solubility. Remember that amino acids have different 'R' groups. These groups can add to the amino acid's polarity and size. For example alanine, whose 'R' group is a -CH3, is much smaller and less polar than say aspartic acid, whose R group is -CH2COOH. Paper chromatography separates components based on their size and polarity. Molecules that are lighter move further up the paper than heavier molecules. Also, since the solvent is polar, more polar molecules will move farther down the paper. | |
Example 1: Identify the amino acid in a chromatogram by calculating their Rf values.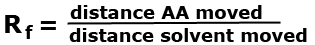 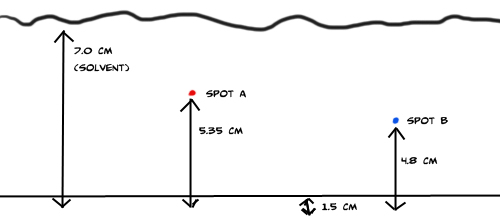 Answer: click to reveal |
Glossary
Amino Acid = an amphoteric organic acid containing the amino group NH2;especially any of the alpha-amino acids that are the chief components of proteins and are synthesized by living cells or are obtained as essential components of the diet .
Protein = any of numerous naturally occurring extremely complex substances that consist of amino-acid residues joined by peptide bonds, contain the elements carbon, hydrogen, nitrogen, oxygen, usually sulfur, and occasionally other elements (as phosphorus or iron), and include many essential biological compounds (as enzymes, hormones, or immunoglobulins).
Precursor = a substance, cell, or cellular component from which another substance, cell, or cellular component is formed.
Hormone = a product of living cells that circulates in body fluids or sap and produces a specific effect on the activity of cells remote from its point of origin; especially :one exerting a stimulatory effect on a cellular activity.
Neurotransmitter = a substance (as norepinephrine or acetylcholine) that transmits nerve impulses across a synapse.
Carboxyl = a univalent radical COOH typical of organic acids -- called also carboxyl group.
Zwitterion = a dipolar ion.
Amphoteric = partly one and partly the other; specifically :capable of reacting chemically either as an acid or as a base.
Chromatography = a process in which a chemical mixture carried by a liquid or gas is separated into components as a result of differential distribution of the solutes as they flow around or over a stationary liquid or solid phase.
Related Materials
For more on amino acids:
The 20 Amino Acids (http://www.people.virginia.edu/~rjh9u/aminacid.html)
For more on paper chromatography:
Paper Chromatography (http://www.allsands.com/Science/paperchromatogr_xqw_gn.htm)